 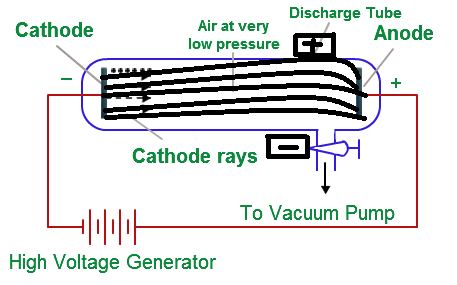 
Cathode rays are deflected by both magnetic and electric fields. Image used with Permission (CC BY-SA-NC). When a high voltage is applied to a gas contained at low pressure in a gas discharge tube, electricity flows through the gas, and energy is emitted in the form of light. The results of these experiments helped Thomson determine the mass-to-charge ratio of the cathode ray particles, which led to a fascinating discovery - minus the mass of each particle was much, much smaller than that of any known atom. THE SMALLEST AND LIGHTEST POSITIVE ION WAS OBTAINED FROM HYDROGEN AND WAS CALLED PROTON. The cathode ray discharge tube experiment does not lead to the discovery of radioactivity. 1: A Gas Discharge Tube Producing Cathode Rays. Thomson also placed two magnets on either side of the tube, and observed that this magnetic field also deflected the cathode ray. The behavior of these particles in a magnetic or electric field is opposite to that of electrons or cathode rays. Some positively charged particles carry a multiple of a fundamental unit of electric charge.Ĥ. 
The charge to mass ratio of particles depends on the gas from which it originates.ģ. Those are simply the positively charged gaseous ions.Ģ. He observed that a beam of particles, called cathode rays, was emitted from the negatively charged cathode and traveled towards the positively charged anode. The positively charged particles depend upon the nature of gas present in the cathode ray tube. The Cathode ray experiment is based on conduction of electricity through gases at low pressure in a discharge tube. The experiment for canal rays was carried out in modified cathode ray tube, by E. the cathode rays consist of electrons, while the anode/canal rays are the positively charged gaseous ions. Thomson theorized that the traces of gas remaining in the tube were being turned into an electrical conductor by the cathode rays themselves, and managed to. You are right, both kinds of rays are emitted simultaneously. The gold foil was surrounded by a detector screen that would flash when hit with an α \alpha α alpha particle. Most of the radiation was absorbed by the lead, but a thin beam of α \alpha α alpha particles escaped out of the pinhole in the direction of the gold foil. In this case, Rutherford placed a sample of radium (a radioactive metal) inside a lead box with a small pinhole in it. Alpha particles are helium nuclei ( 2 4 He 2 ) (_2^4\text) ( 2 4 He 2 ) left parenthesis, start subscript, 2, end subscript, start superscript, 4, end superscript, start text, H, e, end text, start superscript, 2, plus, end superscript, right parenthesis, and they are given off in various radioactive decay processes. Thomson had an inkling that the rays emitted from the electron gun were inseparable from the latent charge, and decided to try and prove this by using a. In his famous gold foil experiment, Rutherford fired a thin beam of α \alpha α alpha particles (pronounced alpha particles) at a very thin sheet of pure gold. The next groundbreaking experiment in the history of the atom was performed by Ernest Rutherford, a physicist from New Zealand who spent most of his career in England and Canada. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
